Contributing Writers & Artists
The Bucky Ball
Contributing Writers & Artists
The Bucky Ball
Introduction
By Thomas T.K. Zung
In 1996, three chemists, Dr. Harold W. Kroto from Great Britain’s University of Sussex, and Dr. Robert F. Curl and Dr. Richard E. Smalley from Rice University in Houston, Texas, were awarded the Nobel Prize in Chemistry. They made their discovery by zapping graphite with a laser beam and mixing the resulting carbon vapors with a stream of helium. They found the molecules made up of sixty carbon atoms arranged like a soccer ball in a geodesic dome sphere that resembled R. Buckminster Fuller’s well-known geodesic dome. At Kroto’s suggestion, they graciously named their discovery, C60’“buckminsterfullerene” – the “buckyball” for short.Their work opened an entirely new branch of chemistry and gave scientists a greater understanding of how nature bonds carbon atoms together.
Robert F. Curl wrote to me after notification of the trio’s Nobel Prize, “I have never had the pleasure of meeting R. Buckminster Fuller in person, but no aware person could live for decades in the twentieth century without meeting him many times in his work.”
Sir Harold W. Kroto, in addition to being a co-recipient of the 1996 Nobel, has received the Longstaff medal of the Royal Society of Chemistry, the Hewlett Packard Europhysics Prize, and the Moët Hennessy/Louis Vuitton Science pour l’Art Prize. He has spoken at the Buckminster Fuller Institute in California.
Macro-, Micro-, and Nano-scale Engineering
By Sir Harold W. Kroto
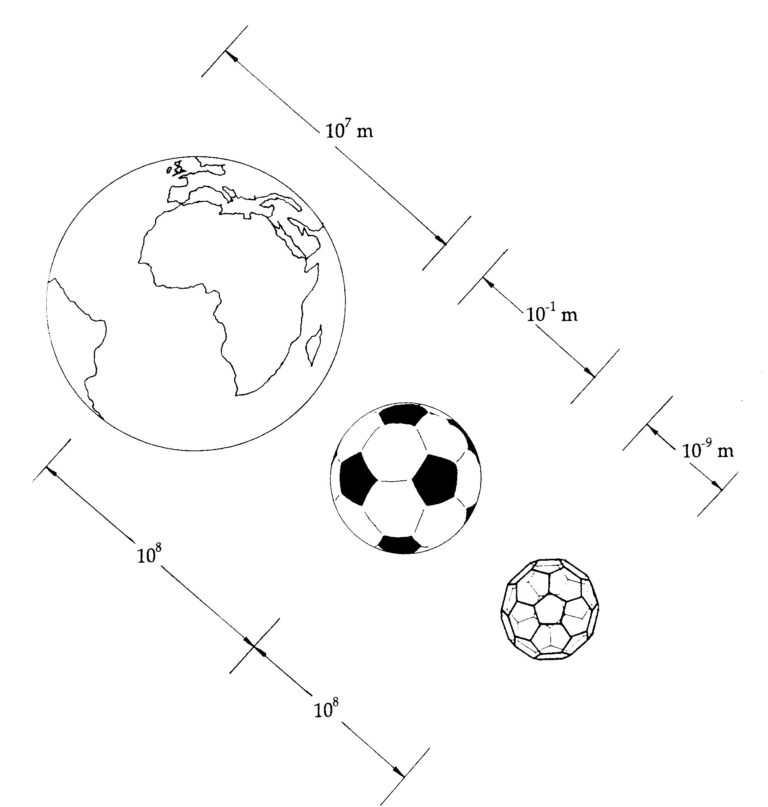
Meccano Magazine, in 1928, printed a letter from Mr. A.H. Finlay of Holyrood, County Down, in Ireland (Fig. 1) in which a truncated icosahedral cage constructed from Meccano pieces (Meccano is known as Erector Set in the United States) was depicted.1 The legend suggested that the structure might be useful as a lampshade – but not much good as a football! A curious error, as this shape has subsequently become the standard pattern for the construction of soccer balls. This letter also makes fascinating reading in light of the development of Buckminster Fuller’s geodesic domes. (Fig. 2), the discovery of C60, buckminsterfullerene2 (Fig. 3). Jonathan Hare – who while working with me extracted C60 (essentially simultaneously with the breakthrough of Kraetschmer, Lamb, Fostiropoulos, and Huffman3 during some excitingly fraught weeks in September 1990 – has pointed out that the world is about 100 million times larger than a soccer ball and a soccer ball is about 100 million times larger than a C60 molecule – i.e., the buckyball (Fig. 4). The well-known drawings of Haeckel of siliceous sea creatures, such as that of Aulonia hexagona (Fig. 5), which appear in the wonderful book On Growth and Form by D’Arcy Thompson,4 led David Jones in 1967 to suggest that carbon might be coerced into forming graphite balloons.5 Furthermore in 1970, while watching his son playing with a soccer ball, Eiji Osawa6 came to the equally imaginative conclusion that the carbon cage C60 molecule with the truncated soccer-ball structure that Bob Curl, Jim Heath, Yuan Liu, Sean O’Brien, Rick Smalley, and I discovered in 19857 would be stable – if it could be made. A year later, in 1971, Osawa, together with Yoshida,8 wrote about this molecule in a book on aromaticity in chemistry. As the geodesic dome images, such as that in Fig. 2, were an important clue to the likely structure of C60, I suggested that we name the molecule after Buckminster Fuller, and after a little struggle they agreed. If we also add to the potpourri the studies by artists such as Leonardo da Vinci and Piero della Francesca (Fig. 6), we see that this amazingly charismatic framework has been a geometric force underlying the creativity of a wide range of people as well as numerous natural and physical processes.
Click images to view them larger.
As we enter the new millennium, it is worth throwing all these observations in a (think) tank, stirring them all up together, and ruminating (hopefully imaginatively?) about the possible implications (hopefully beneficial! – this was, after all, the pattern of the charges used to “detonate” the atomic bomb) on the fact that the icosahedral structure has now surfaced as a ubiquitous unifying geometric “engineering” principle in the nanoscale world. Of course, the C60 molecule probably first formed on Earth when the forest fires around since time immemorial, since carbon atoms (mass 12) – now in our bodies – were synthesized by the curious triple-alpha process in stars. In this process gravitational attraction caused three He nuclei (mass 4; 3x 4=12) to fuse together in carbon stars, which then exploded as red giants blasting carbon out into space as atoms, polyyne chain molecules, carbonaceous soot like dust, and almost certainly also C60 as well as other fullerenes. C60 is, however, probably not in high enough abundance to be definitely detectable at this time. The C atoms and molecules then wafted around in space to end up by some “Umberto Eco-type” chance on our Earth – some in our bodies, yours and mine, and unfortunately also in the bodies of some others whose atoms should, in my opinion, still be waiting about in space.
The term “engineering” tends to evoke the names of those giants of yesteryear who created the civil engineering structures that transformed our world – Isambard Kingdom Brunel and Richard Trevithick and James Watt and the Wright Brothers. Today, however, the term embraces a far wider range and covers even more fascinating areas. It has expanded to cover architectural constructions such as the amazing Sagrada Familia of Antonio Gaudí, Frank Lloyd Wright’s Marin County Civic Center, and Norman Foster’s Hong Kong Shanghai Bank. Perhaps no other building combines the fundamental of structural design and architecture more organically than does the geodesic dome of R. Buckminster Fuller. Perhaps it epitomizes Lloyd Wright’s axiom that “form and function are one” even more purely than any of Lloyd Wright’s creations. On top of all this we should not forget that, as we reach the end of the twentieth century, similar intellectual processes are involved in the creation of microchips – those tiny constructs that are revolutionizing our lives. Under the microscope they are seen to be elegant abstract mosaics, providing some of the most elegant art forms of the twentieth century.
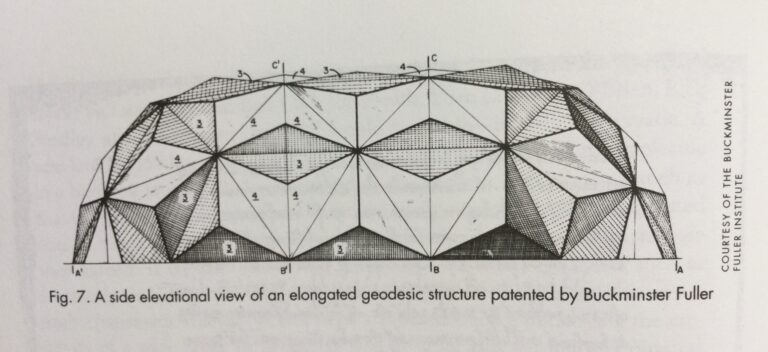
For all its excitement, our discovery of C60 was most important because it showed that sheet materials on an atomic/molecular scale spontaneously form closed cages. Almost exactly five hundred years after 1492, we have come full circle and, just as Columbus explored a New “Round” World, we can now explore a new aspect of carbon chemistry only to discover a new round world of sheet materials. We should, however, have known about it much earlier – at least in the 1960s – as some clues were being uncovered by the combustion community, which had already recognized that some soots and carbon blacks consisted of grapheme sheets organized in roughly concentric shells. What is more, some carbon fiber studies had revealed structures that were thought to be Swiss-role structures rather than the concentric carbon nanotubes (buckytubes) that we now recognize them to be today. These amazing new materials hark back to the patents of Buckminster Fuller9 in which the elongated geodesic structures diagrammed (Fig. 7) are essentially macroscopic prototypes of C70 and the buckytubes first recognized by Sumio Iijima.10 These structures are not just restricted to carbon, as other multiple sheet materials such as molybedenum sulphide – as shown by Tenne and coworkers11 – also can form related structure (Fig. 8).
Click images to view them larger.
So now we have the tantalizing glimpse of a new material, which could revolutionize civil and electronic engineering much as steel and aluminum did in earlier times. The buckytube discovery promises a material some fifty to one hundred times stronger than steel at about one-sixth the weight, a material which could provide nanometer-diameter, low-resistance wires to interconnect the nanoscale components on the molecular computer chips that appear to be just over the horizon. However, to harness the exceptional tensile strengths as well as the electronic and magnetic properties of these molecular structures, advances in synthetic technology that rival the ingenuity breakthroughs of the past will be required. We need molecular engineering with a precision as yet well beyond us. It will require us to leap forward from the past approach in which chess-type logic was used to outmaneuver entropy and create new, ever more complex molecules. This was the board on which such wizards of synthetic chemistry as Robert Burns Woodward, Robert Robinson, and John Cornforth played an on which their heirs such as Jean Marie Lehn, K.T. Nicolau, and Fraser Stoddart are playing today.
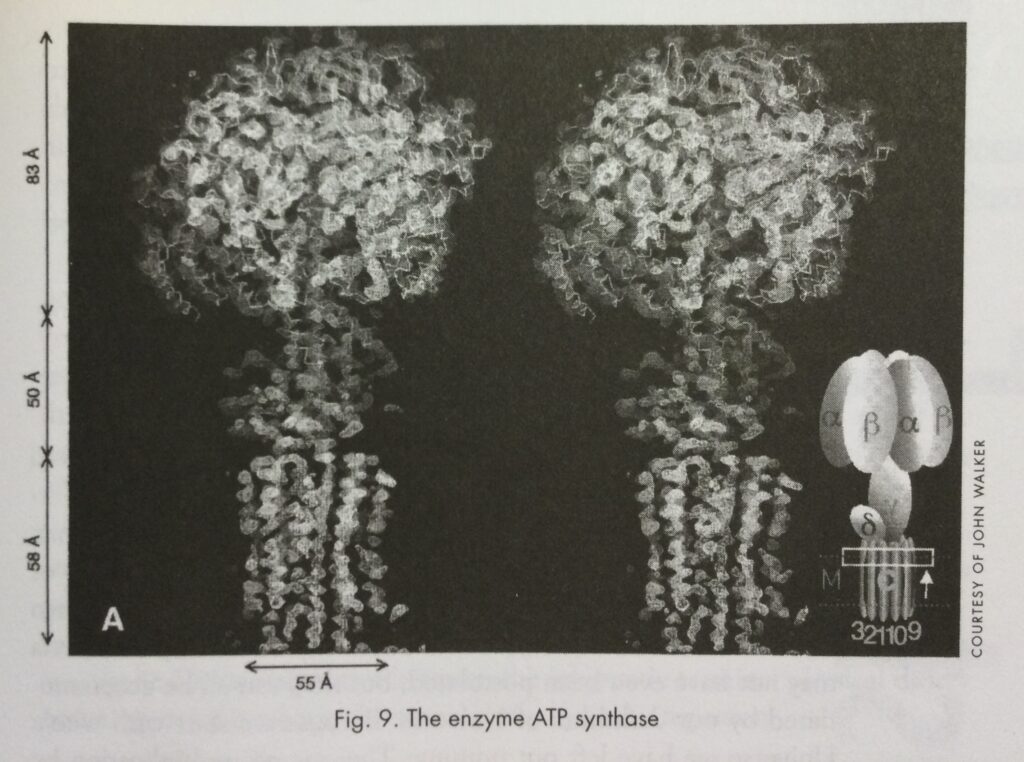
Perhaps we should take a leaf out of the Natural World’s book and attempt to create the nanoscale jigs that will allow us to hold in place the components of the nanoscale (pure carbon) Meccano components we have discovered, so that we can build truly complex, exactly specified giant molecules. To achieve its aims, “Life” has invented proteinaceous enzymes which are effectively gantries, much like scaffolding we use to build the skyscrapers of today. A particularly delightful example, which exists in our bodies, in the amazing enzyme ATP synthase (Fig. 9) of Paul Boyer (UCLA) and John Walker (MRC Cambridge), who were awarded the 1997 Nobel Chemistry Prize for elucidating its structure and mode of action.12 It seems, at least to me, to be a portent of fascinating chemistry of the future. The advance has similarities with the breakthrough made by Max Perutz, who discovered that deoxyhemoglobin (another favorite of mine) had developed the knack of readjusting its structure on oxygen uptake in order to facilitate further oxygen uptake (up to four molecules). This is really unusual, as in general one would expect oxygen uptake to get harder as successive molecules are added – as in the case of when one blows up a balloon which gets more and more difficult. Boyer and Walker found that there are tiny biological electric motors in our bodies which are essentially molecular machines consisting of a segmented pumpkin-shaped protein structure with an axial central shaft. As an asymmetric rotor in the shaft turns, driven by a flow of H ions through a disc at its base, it distorts cavities in the walls of three of the segments. The motion enables this amazing molecular machine to drive the addition of phosphate (P) to adenosine diphosphate (ADP) to form adenosine triphosphate (ATP) in the cavities.13 In muscle tissue the ATP can revert back to ADP + P, and the energy stored in ATP is released, enabling us to move our arms and legs. What an amazing delight it is to discover that our muscles are powered by myriads of microscopic electric motors. We turn over at least half our body weight of ATP each day. The more we learn about nature, the more we seem to find that we are reinventing the wheel – or in this case the armature.
Perhaps a new age of chemistry – truly nanometer-scale molecular engineering – is on the horizon, an age in which we still start by modifying existing enzymes to make new molecules perfectly. New advances in genetic engineering will be required. Perhaps in the next stage we will learn how to build our own enzyme-like gantries – perhaps mainly out of carbon – and how to perfect a synthetic chemistry technology capable of rivaling the staggering efficiency of DNA replication (error rate of 1:109). Any higher error rate than 1:109 for DNA would be too high for us to exist for long enough! Such synthetic strategies seem a far cry from our present capabilities. Hopefully, however, now that we know what is achievable, the young geniuses of the new millennium will develop such techniques, spurred on by aesthetic visions of the novel molecular architecture which nature has created.
Acknowledgements: I wish to thank my colleagues Jonathan Hare, Humberto Terrones, and David Walton for their help with this article.
Footnotes
1. Incidentally, I lay the blame for the death of engineers and scientists in the West fairly and squarely on the fact that Lego has replaced Meccano as a “toy” of choice in many homes in the developed world. I use the word “toy” advisedly, because Lego is a toy, whereas Meccano is not only a toy but also an amazing introduction to the world of real engineering. It gives the child a real understanding of how nuts and bolts should be put together. They learn how to tighten a nut and bolt sufficiently tightly to not misthread it, and they get a feel for materials and learn how metal structures actually work. (An anecdote: A retired gentleman, who kindly came to display his amazing pantographs to young children at the 1999 BA festival in Sheffield, told me that he made a four-speed gearbox when he was eleven, and when he got his first job, at twelve, he was essentially already trained for it.)
2. H. W. Kroto, J. R. Heath, S. C. O’Brien, R. F. Curl, R. E. Smalley, Nature (London) 1985, 318, 162-163
3. W. Kraetschmer, L. D. Lamb, K. Fostiropoulus, D. R. Huffman, Nature (London), 1990, 347, 254-358.
4. D”A. W. Thompson, On Growth and Form (Cambridge: Cambridge University Press, 1942).
5. D. E. H. Jones, New Sci. 32 (3 November 1966) 245; D. E. H. Jones, The Inventions of Daedalus (Oxford: Freeman, 1982) 118-119.
6. E. Osawa, Kagaku (Kyoto) 1970, 25, 854-863 (in Japanese); Chem. Abstr. 1971, 74, 75698v.
7. Kroto, Heath, O’Brien, Curt, Smalley, op. cit.
8. Z. Yoshida, E. Osawa, Aromaticity (Kyoto: Kagakudojin, 1971) 174-178 (in Japanese)
9. R. B. Fuller, Inventions – The Patented Works of Buckminster Fuller (New York: St. Martin’s Press, 1983).
10. S. Iijima, Nature (London) 1991, 354, 58.
11. R. Tenne, L. Margulis, M. Genut, G. Hodes, Nature, 1992, 360, 444.
12. D. Voet, J. G. Voet, C. W. Pratt, Fundamentals of Biochemistry (New York, Wiley, 1999) 514-517.
13. Voet, Voet, Pratt, op. cit.
Fullerenes, Nanotubes and Nobel Prizes: 25 years of C60
By Dr. Jonathan Hare
The Creative Science Center, Sussex University
This article was previously published in The Ethical Record, Vol. 115, No.8, in September 2010 , and appears here with permission from the author.
Summary
In 1985 the science world was shaken by a new discovery that has, and is still transforming our understanding of carbon. What follows is a brief account of the discovery of a family of carbon cage molecules called the Fullerenes – the third allotropic form of carbon. In 1996 the chemistry Nobel Prize was awarded for its discovery. This story is an exciting development in the history of science and a vibrant new area of nanotechnology.


The structure of buckminsterfullerene (C60) and the common soccer ball.
Latest News
Since going to press some new results have been published: C60 and the fullerenes were discovered on Earth as a unexpected result of experiment trying to mimic chemistry that might be going on in the material produced from stars and possibly within molecular clouds in the inter stellar medium. On the 22nd July 2010 scientists revealed that C60 has finally been detected in the ejected material from a star 6500 light years away.
See the BBC News article here.Introduction
Carbon must rate as one of the most studied of the 92 naturally occurring chemical elements. Yet it was only in 1985 that completely new forms of pure carbon were unexpectedly discovered1-9. These are the Fullerenes; Buckminsterfullerene (also called C60) being the head of these fascinating new structures1. In 1996 the Nobel Prize for chemistry was awarded to Kroto, Curl and Smalley for the discovery7. I was lucky enough to be involved in some of this pioneering work during my PhD studies8.
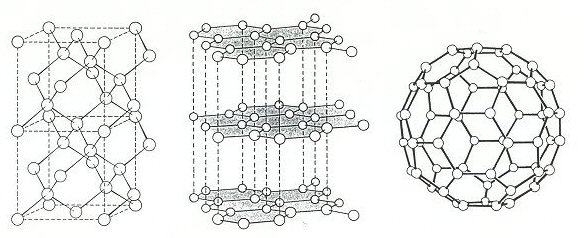
Three allotropes of carbon: (l to r) diamond, graphite and C60 (a fullerene).
Two allotropes: diamond and graphite
Diamond and graphite are the pure forms of carbon that we learn about early on in science education – they are the two allotropes of carbon. They are classic examples of how the same atoms can have very different properties when combined together depending on the way the atoms are arranged – i.e. their structure. In diamond each atom is joined to four neighbouring atoms in a 3D tetrahedral ‘scaffolding’ type structure which accounts for its great strength. In graphite the atoms have three neighbours forming predominantly flat 2D hexagonal sheet networks. In graphite the sheets lie one upon the other and are fairly weakly bound together and so easily part, making graphite soft and lubricating. They get their names from the ancient Greek; diamond meaning ‘invincible’ – the hardest naturally occurring material, while graphite means to ‘write’ it’s still used today in pencil ‘leads’. Bearing in mind the wide use of carbon and its importance to humans throughout the ages it’s very surprising that it was only relatively recently (1985) that another, third form, of carbon was discovered.
The Carbonaceous Component of the Cosmos
In a paper we wrote in 1992 Harry Kroto and I outlined the wide spread and vital role carbon plays in the universe9. Astronomy has played two key roles in the discovery and development of the Fullerenes; in fact without this ‘blue sky’ research the fullerenes might still be awaiting discovery.
Astrochemisty
In the 1970’s Harry Kroto and David Walton (both at Sussex University, UK) ran an interesting collaborative research programme investigating radio wave emissions from carbon based molecules3,4,7. In particular they studied straight chain carbon molecules e.g.:
H-C≡C-C≡N or more simply HC3N
H-C≡C-C≡C-C≡C-C≡N or more simply HC7N
Two linear chain carbon molecules
When these molecules tumble and rotate in a gas (because of the thermal energy) they often radiate energy at the same frequency as they rotate. This ‘rotational emission’ is in the radio wave (microwave) part of the electromagnetic spectrum. David Walton and his students would make the molecules while Harry Kroto and colleagues would measure their corresponding microwave spectrum. This data would be given to the radio astronomers who would turn their antennas and receivers to carbon rich objects in the universe to try and spot their tale-tale signs. Not only were many of the laboratory molecules observed but they were in great abundance in the vast molecular clouds in space.
The emission spectra can be used to estimate the abundance and average temperatures within these clouds. The molecular microwave emissions are crucial in allowing energy to escape as gas clouds (gravitationally) collapse. It allows the heating effect of compression to dissipate eventually forming conditions dense enough for stars to form. Dense molecular clouds can be the birthplace of stars.
The 1985 cluster beam experiment
In order to try and understand why carbon molecules were so abundant in space Harry Kroto (at the suggestion of Bob Curl, Rice University, Texas) joined up with Rick Smalley (also Texas) to use his ‘cluster beam’ apparatus.
Smalley had pioneered the investigation of small clusters of atoms and to Kroto his technique seemed ideal for astrochemical investigations of carbon1,3,4. His apparatus consisted of a large vacuum chamber within which materials could be vaporised using a high power laser. In practice this laser experiment produced only tiny amounts of the interesting product but just as importantly it created ‘tones’ of new data, in particular the mass spectrum of new things. The laser heated the samples to 10,000°C (similar to the surface temperatures of a star) and the products were swept into an interstellar space-like vacuum chamber. Therefore the laboratory apparatus was an interesting analogue of processes occurring in space. When graphite was introduced into the apparatus it was immediately apparent that the results were very interesting. Below 20 carbon atoms the data seemed to fit in with was known in space but above 20 atoms, and especially from 60 carbon atoms onward, it appeared that something new had accidentally been uncovered1. The peak at 60 carbon atoms – the C60 molecule – seemed very special indeed as it tended to dominate the data. Remember that what they were vaporising was graphite – sheets of atoms arranged in hexagons. On heating up by the laser these atoms appeared to be (spontaneously) rearranging to form all sorts of interesting structures. It was proposed that the graphite sheets might be curling up to form cages, which being closed-up might account for their stability. In their efforts to understand what was going on in these experiments the scientists remembered the architectural structures of Richard Buckminster Fuller.
Richard Buckminster Fuller (Bucky) and the Geodesic dome
Buckminster Fuller’s life’s work explored structures and he is most famous for the geodesic dome. He was interested in the structures and principles that emerge spontaneously as things are brought together10,11 – synergy:
Synergy is one
of those generalised principles
it is defined scientifically
as behaviour of whole systems
unpredicted by behaviours
of any of their separate parts
—Intuition, R.B. Fuller 11
A famous example of RB Fuller’s work includes the enormous Expo 67 dome in Montreal, Canada10. He inspired architectural buildings such as the Epcot center in Disneyworld (Florida, USA) and the Eden project (Cornwall, UK). Studying these structures they look like they are composed of hexagons (often the hexagons are subdivided into 6 regular triangles). Looking down on a dome you see that at the top there is also a pentagon (also triangulated but with irregular triangles) followed by five other pentagons around the base. For a dome 6 pentagons are required. Two domes joined to form a cage or sphere would require 12 pentagons.

The author with a small home-made geodesic dome composed of wooden dowels.
Once the scientists noticed that Fuller incorporated pentagons into his structures they started to include pentagons into their ‘hexagonal’ graphite models. Each time a pentagon was introduced it curved the hexagon sheet a little, until with 12 pentagons the sheet closes up into a cage, or ball. If we separate every pentagon by a single edge a particularly elegant solution appears – it’s a football ! (more accurately called the truncated icosahedron1,3,4 see diagram).
Now as each of the 12 pentagons has five corners and there are no corners not in pentagons (take a long look at a football) then we must have 12 x 5 = 60 corners or vertices. Exactly the number that seemed so special in the experiment. Suddenly they could propose a structure for C60. The fact that pentagons are essential for closure was crucial in the structural solution of the football C60 proposal and so Harry Kroto named the molecule Buckminsterfullerene, or Buckyball in honour of the architect.1,3,4
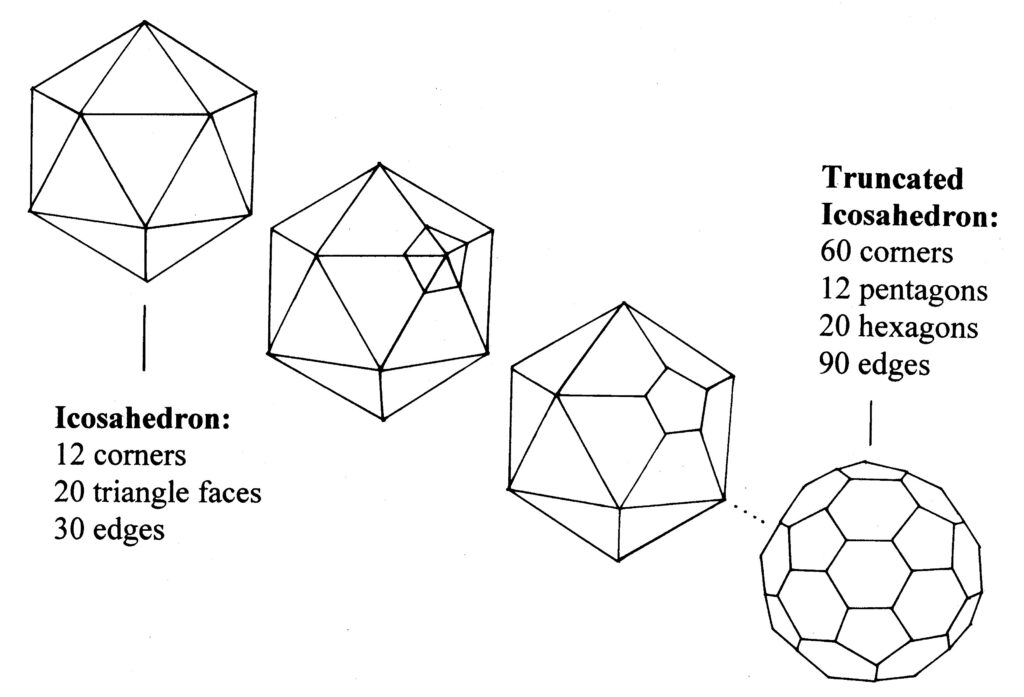
Truncating the icosahedron- the structure of a football.
The carbon arc
Not only are there vast hoards of molecules in interstellar clouds but these clouds also contain particles which astronomers call ‘dust’. In the 1990’s several groups were performing laboratory experiments to try and understand this dust in detail by making carbon dust by electrically arching carbon rods. While working in Harry Kroto’s group, I and others developed a way to make large (large to a chemist) gram quantities of the fullerenes3,4,9 during arcing within He or Ar gas at reduced pressure (1/10th atmospheric pressure). The black soot produced can be up to 10% (by weight) C60. The molecules can be solvent extracted (toluene, hexane etc.) for purification and for performing chemical reactions8.
The post Buckminsterfullerene era
A buzz word of modern technology is nanotechnology. These are structures a few billionth of a meter in size i.e. several nanometers (few nm) – hence ‘nanotechnology’. The fullerenes are nm’s in diameter so are therefore at the heart of a new branch of nanotechnology. Fullerenes can also form into tubes called nanotubes3,4,6,14,15. These are a few nanometers in diameter (hence their name) but can be relatively very long. The tubes can be insulating, semiconducting or conducting (metallic) depending on their diameter. Hence it’s possible to envisage a nano-cable made of a tiny conducting inner nanotube surrounded by a slightly larger diameter insulating outer carbon nanotube (CNT). Estimates suggest that tubes may be 100 times the strength of steel but a 1/6th of the weight! Recently a single CNT was used as a resonant detector, tuner and amplifier in the world’s smallest FM radio.
Blue sky research
It is interesting to note that both the discovery of C60 (ca. 1985; molecules in space) and the method of production (ca. 1990; dust in space) students played a very important part in the developments3,4,8. This was partly because the researchers were positively displaced towards using students for ‘frontier research’ and partly because such curiosity driven endeavours are hard to get funding for. Students often came with bursaries (British Gas in my case) which helps get interesting and unusual investigations started quickly without the bureaucracy and red tape of additional fund raising. Most crucially as the work develops students are adaptable. A very focused research group would not be nearly so versatile.
C60 and schools – the great game
Since 1990 Harry Kroto and I have been presenting 100’s of talks and workshops to schools, colleges and science festivals12. Recently with the Vega Science Trust (Gill Watson and Harry Kroto) we have made a series of mini-films for teacher, pupils and students explaining fullerenes in more detail as well as other areas of science13,14. The jump in size from the Earth to a football is about the same leap as from a football to a molecule of C60. Being about a nanometer in diameter, C60 is therefore a wonderfully simple way of appreciating the fascinating new world of fullerene nanotechnology.
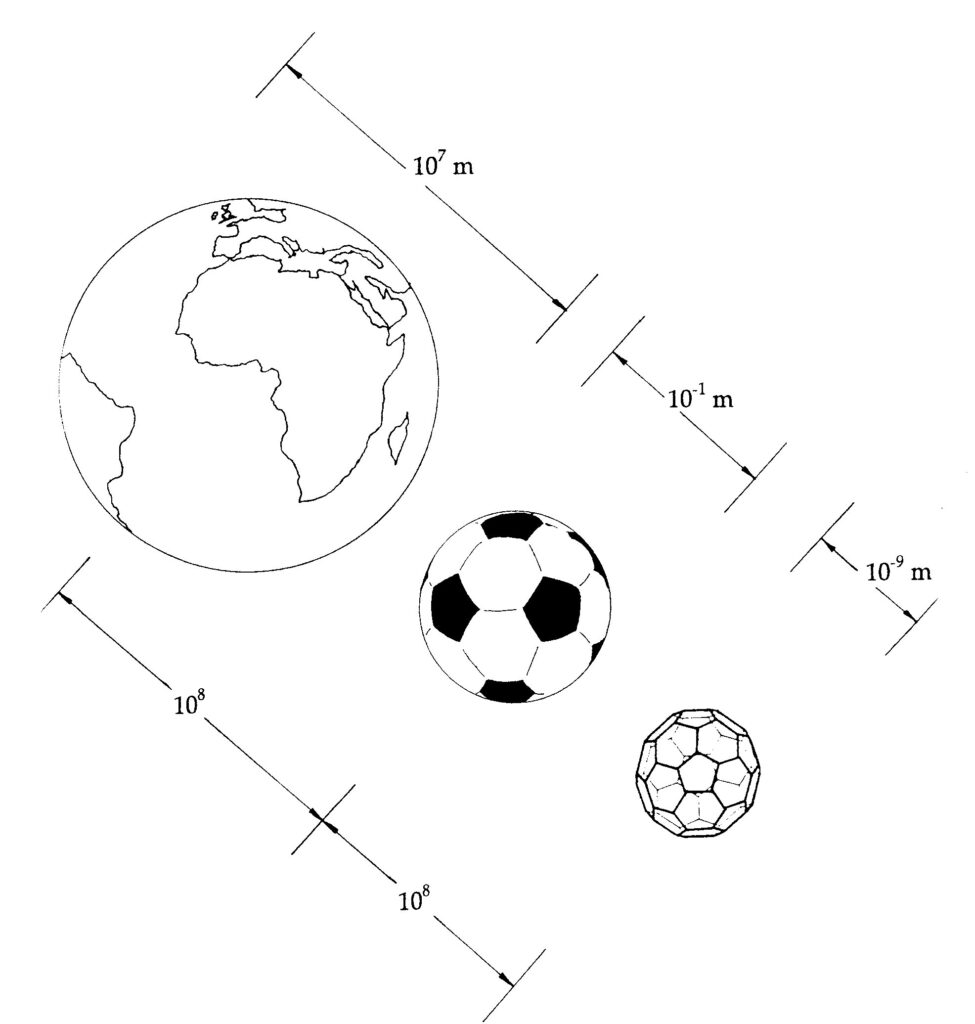
A football is as many times smaller than the Earth as a C60 molecule is to a football. A jump of about 100,000,000 times. 8
Footnotes
1. Discovery of C60, HW Kroto, JR Heath, SC O’Brien, RF Curl, RE Smalley, Nature, 1985, 318, 162.
2. R Taylor, JP Hare, AK Abdul-Sada, HW Kroto, Journal Chem. Soc. Chem. Communication. 1992, 1423
3. Perfect Symmetry, Jim Baggot, Oxford, 1994. ISBN 0 19 8557906
4. The Most Beautiful Molecule, Hugh Aldersly Williams, Aurum Press, 1994. ISBN 1 85410 303 2.
5. Designing the molecular world, Philip Ball, Princiton University Press, 1994. ISBN 0 691 000581.
6. Molecules with sunglasses Horizon, BBC TV, 1992.
7. 1996 Nobel Prize for Chemistry; Fullerenes
8. C60, Buckminsterfullerene; extraction, characterisation and astrophysical implications. JP Hare PhD thesis, Sussex University, 1993.
9. A Postbuckminsterfullerene view of carbon in the galaxy, JP Hare and HW Kroto, Accounts of Chemical Research, 1992, 25, 106-112.
10. Design Heroes: Buckminster Fuller, Martin Pawley, Harper Collins, 1990. ISBN 0 586 08881 4
11. Intuition, R B Fuller, Doubleday, 1972
12. see CSC web site
Digizyme
By Gael McGill, Ph.D.
Cellular Landscaoe
A eukaryotic human cell represents one the most complex units of life ever studied and contains well over 1 billion proteins. The exact choreography of these molecular actors remains one of the most intriguing and intensely studied areas of biology. The cellular landscape depicted below is an attempt to capture this dizzying complexity, albeit in a very simplified static form. Created by Evan Ingersoll and Gaël McGill for Cell Signaling Technology, Inc., the image is a prime example of the scientific visualization work carried out by Digizyme, the scientific visualization company founded by Gaël McGill in 1999.
Inspired by the stunning watercolor art of scientist-artist David Goodsell, this computer-generated 3D rendering is modeled based on molecular structure coordinates derived from X-ray, nuclear magnetic resonance (NMR), and cryo-electron microscopy datasets in the Protein Data Bank. Although very dilute in its concentration relative to a real cell (where molecules entirely fill available space), the image is also an attempt at recapitulating the myriad pathways involved in signal transduction, protein synthesis, endocytosis, vesicular transport, cell-cell adhesion, apoptosis, and other processes.
Closer inspection of this landscape reveals structures reminiscent of Buckminster Fuller’s geodesic domes and C60 fullerenes: clathrin-coated vesicles that form similar soccer-ball shaped structures (albeit at a scale roughly 100 times larger than a buckyball and 1000 times smaller than a soccer ball). These clathrin cages, which self-assemble from individual three-legged proteins called triskelions, allow cells to ferry outside materials (be they harmless food particles or viruses!) into the cytoplasmic interior. Interactive versions of parts of this landscape can be explored here.

Thomas T.K. Zung
Writer
Thomas Tse Kwai Zung was born in Shanghai, China. He was a student of Buckminster Fuller. With Fuller’s Synergetics, Inc., he designed the elongated geodesic dome in Cleveland, Ohio, in 1968. He has worked on various geodesic domes, including the Jitterbug sculpture, Tensegrities, the Fly Eye’s dome, and Fuller’s last invention, the Hang-It-All. Zung is president of Buckminster Fuller, Sadao, and Zung, and he also serves as board member of the Buckminster Fuller Institute.

Sir Harold W. Kroto
Writer
From Wikipedia:
Sir Harold Walter Kroto FRS (7 October 1939 – 30 April 2016), known as Harry Kroto, was an English chemist. He shared the 1996 Nobel Prize in Chemistry with Robert Curl and Richard Smalley for their discovery of fullerenes. He was the recipient of many other honors and awards. Kroto held many positions in academia throughout his life, ending his career as the Francis Eppes Professor of Chemistry at Florida State University, which he joined in 2004. Prior to this, he spent approximately 40 years at the University of Sussex.

Dr. Jonathan Hare
Writer
Dr. Jonathan Hare is a freelance science communicator. His PhD work with Sir Harry Kroto found a method of making the football molecule C60, Buckminsterfullerene. He has worked as a ‘Time Lord’ at the National Physical laboratory working with atomic clocks as well as with British Gas developing a gas powered car. He has been on all the BBC Open University Rough Science (6 series) and Hollywood Science (2 series) TV programmes. He is currently a visiting lecturer in science communication in the Physics department at Sussex University.

Gael McGill, Ph.D.
Writer
Gael McGill, Ph.D. is founder and CEO of Digizyme Inc., creator of the Clarafi scientific visualization portal and the Molecular Maya software platform. He is also faculty and Director of Molecular Visualization at the Center for Molecular and Cellular Dynamics at Harvard Medical School where his teaching and research focuses on visualization design methods in science education. He co-authored and was Digital Director of E.O. Wilson’s Life on Earth digital biology textbook for iPad created in partnership with Apple.

Mark Wheeler
Artist (3d Renderings)
Marc J. Wheeler is a designer, assistant art director, and concept artist working in the entertainment industry. Marc has assisted and designed on projects internationally, ranging from theater to opera, to feature film, and to tv shows. Marc completed his BFA at Southern Oregon University, and his MFA at NYU Design for Stage and Film.
Diatribe Diaries
D.S. Legters
The Bucky Ball
Contributing Writers & Artists
Dragă, Sunt Aici Cu Tine
Isabel Mareş